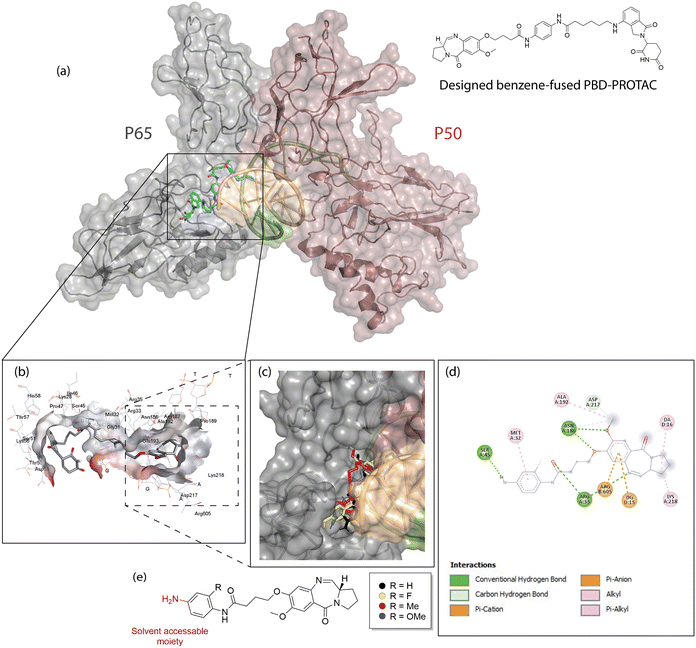
We are pleased to announce that our team has published a landmark study describing the first pyrrolobenzodiazepine (PBD)-based proteolysis-targeting chimera (PROTAC), opening a new therapeutic avenue for cancers driven by dysregulated NF-κB signalling.
The NF-κB pathway is a critical regulator of immune response and cell survival. Its frequent dysregulation in human cancers has made it a long-standing therapeutic target. However, previous attempts to directly inhibit NF-κB or its upstream kinases have been hindered by toxic side effects in normal tissues. Our work provides an innovative alternative—targeted protein degradation of a specific NF-κB subunit.
Key Findings
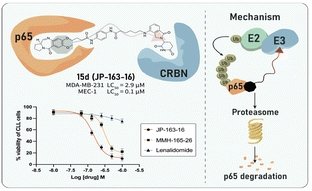
- Selective Degradation of RelA/p65: We developed a C8-linked PBD-PROTAC (JP-163-16, compound 15d) that selectively degrades the NF-κB RelA/p65 subunit in a proteasome-dependent manner.
- Potent Activity in Cancer Cells: Compound 15d showed cytotoxicity with LC50 values of 0.14 μM in MEC-1 cells, 0.23 μM in primary chronic lymphocytic leukaemia (CLL) cells, and 2.9 μM in MDA-MB-231 breast cancer cells.
- Reduced Toxicity in Healthy Cells: In contrast, 15d was two orders of magnitude less toxic in normal B- and T-lymphocytes, indicating a favourable therapeutic window.
- Mechanistic Validation: By linking a C8-PBD to a cereblon-targeting ligand, the PROTAC design abolished DNA binding by the PBD while retaining cancer-selective cytotoxicity via RelA/p65 degradation. Importantly, loss of efficacy in cereblon-deficient myeloma cells confirmed the PROTAC mechanism.
Why It Matters
This is the first reported PROTAC capable of selectively degrading a single NF-κB subunit. By focusing on RelA/p65, this approach has the potential to overcome the limitations of broad NF-κB inhibition and deliver targeted, less toxic therapies for RelA/p65-dependent tumours.