We are proud to announce that our recent work has been published in the Journal of Medicinal Chemistry, the flagship journal in drug discovery. This study addresses one of today’s most pressing global health challenges: the rise of antifungal resistance.
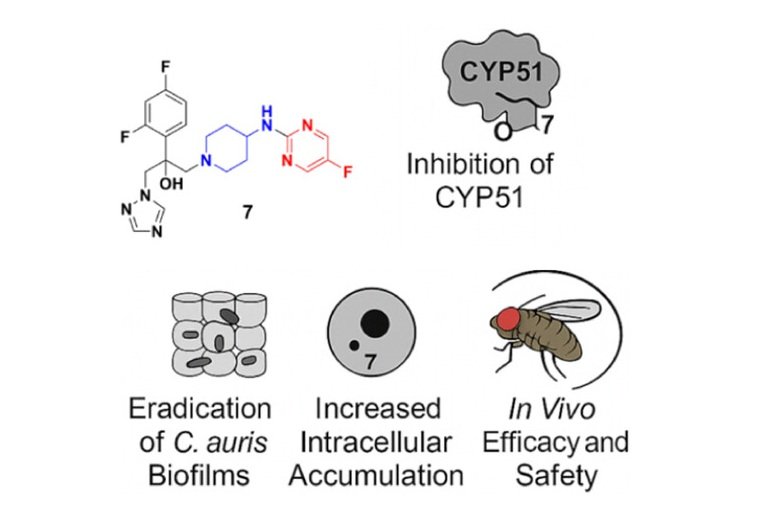
Candida auris has emerged as a dangerous, multidrug-resistant fungal pathogen, responsible for life-threatening infections with very few effective treatment options. To tackle this, our team designed and synthesized a series of novel azole-based compounds with unique cyclic heteroaliphatic linkers bridging aromatic and heteroaromatic rings.
Key Findings
- Broad Antifungal Potency: Several compounds exhibited strong activity against C. auris, including resistant strains, with MICs as low as 0.016 μg/mL. They also showed potent effects against other clinically relevant species such as C. albicans, Nakaseomyces glabratus, C. tropicalis, and C. parapsilosis.
- Enhanced Performance vs. Standard Therapy: Compounds 7, 18, and 21 outperformed the widely used antifungal fluconazole, demonstrating higher potency.
- Mechanistic Insights: Compound 7 inhibited CYP51, eradicated C. auris biofilms, and accumulated more efficiently inside fungal cells compared to fluconazole.
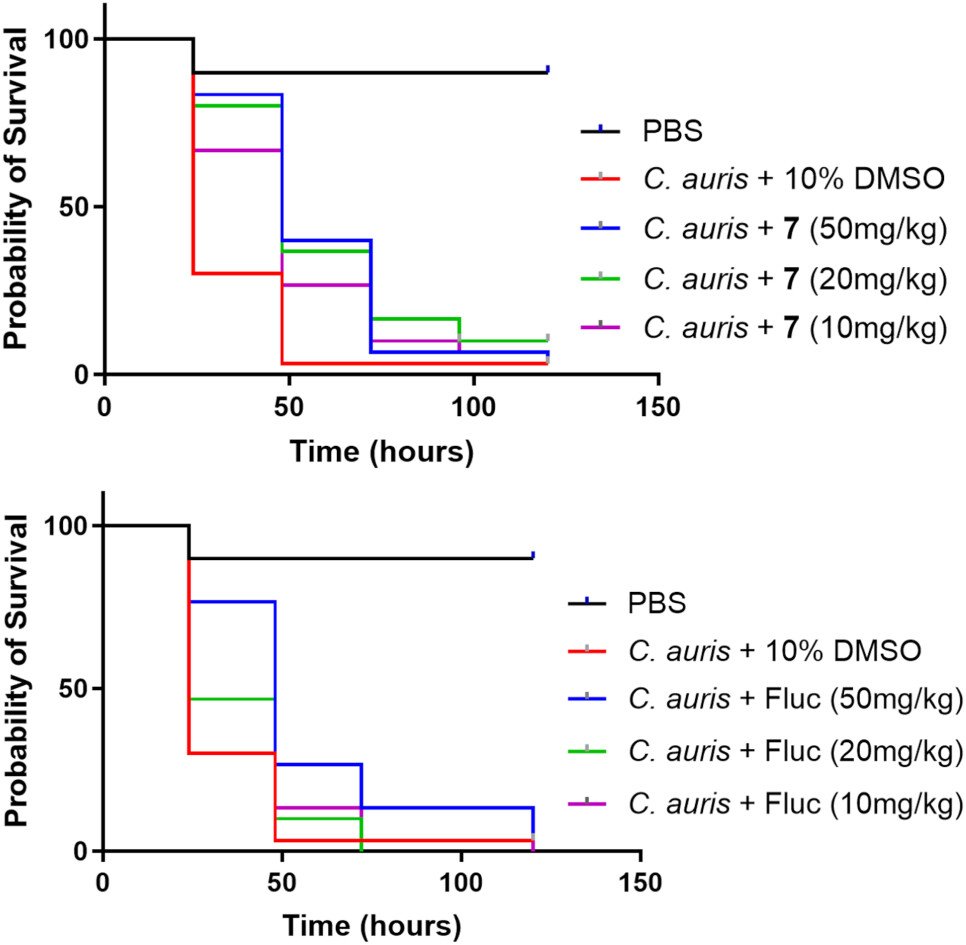
- In Vivo Efficacy & Safety: In animal infection models (Galleria mellonella and Drosophila melanogaster), Compound 7 showed significant efficacy at 5 mg/kg and no toxicity up to 50 mg/kg, supporting its therapeutic potential.
Why This Matters
The discovery of this new azole scaffold opens a promising path for the development of next-generation antifungal agents. With C. auris now classified as a serious global health threat by the CDC and WHO, these findings represent a crucial step toward expanding the arsenal against multidrug-resistant fungal infections.