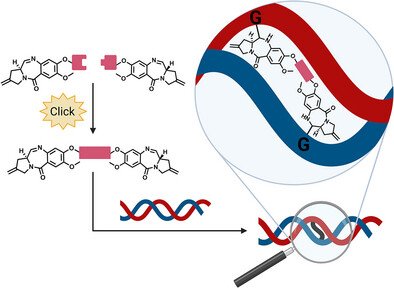
We are delighted to share that our latest research has been published in Chemistry – A European Journal. The study highlights a promising new route to develop pyrrolo[2,1-c][1,4]benzodiazepine (PBD)-based DNA binders using the powerful tools of click chemistry.
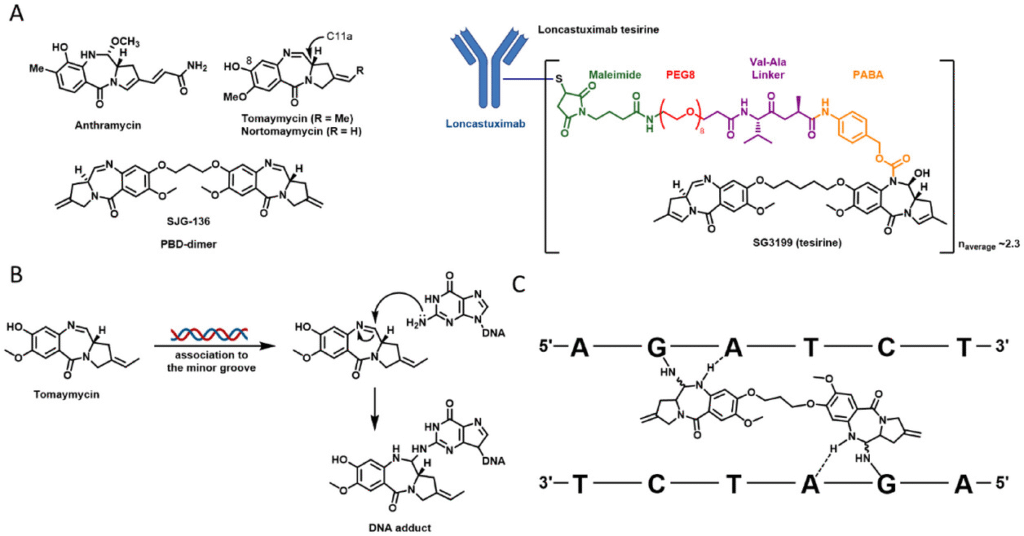
PBDs are a class of small molecules known for their ability to covalently bind and cross-link DNA strands—an established mechanism used in anticancer therapy. They are already deployed as potent payloads in antibody-drug conjugates (ADCs). However, generating diverse and effective PBD dimers has traditionally been a complex process.
In this work, our team demonstrated that copper-catalyzed and strain-promoted azide–alkyne cycloadditions (SPAACs) provide a simple and efficient method to prepare PBD heterodimers.
Key Findings
- DNA Binding & Cross-Linking: Using thermal denaturation analysis, mass spectrometry, and DNase I footprinting, we confirmed strong DNA binding and covalent cross-linking activity of the synthesized dimers.
- Potent Biological Activity: One heterodimer showed remarkable DNA cross-linking and induced strong cytotoxicity in cancer cell lines, underscoring its therapeutic potential.
- Future Applications: The results point to the feasibility of generating PBD dimers inside living cells, opening a pathway for new strategies in targeted drug development.
Why It Matters
This study positions click chemistry as a powerful enabler for rapidly expanding the chemical diversity of PBD-based compounds. The approach not only simplifies synthesis but also creates opportunities for designing next-generation anticancer agents with high precision and potency.
We believe this work provides an important step toward advancing DNA-binding therapeutics and broadening the toolkit for cancer drug discovery.